OUR ROBUST ONCOLOGY PROGRESS
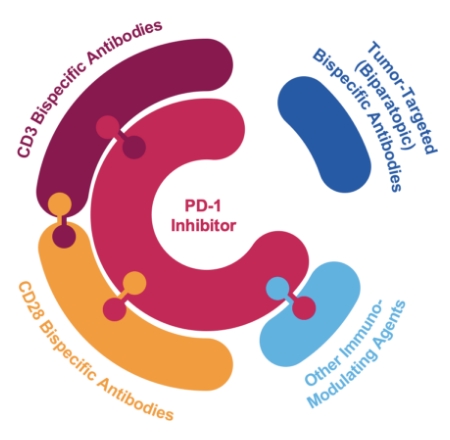
We’re applying more than three decades of scientific innovation with the goal of developing paradigm-changing therapies for patients with cancer. Our oncology portfolio is built around two foundational approaches — our approved PD-1 inhibitor Libtayo® and investigational bispecific antibodies — that are being evaluated both as monotherapies and in combination with emerging therapeutic modalities. Together, they provide us with unique combinatorial flexibility to develop potentially synergistic treatments for a wide range of solid tumors and blood cancers.